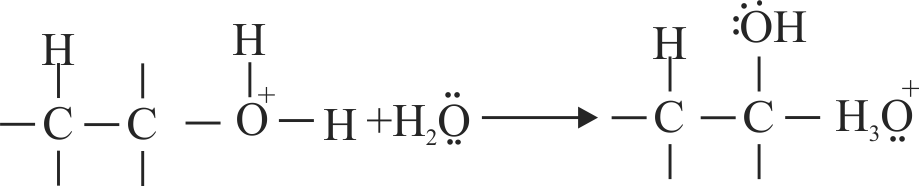322894
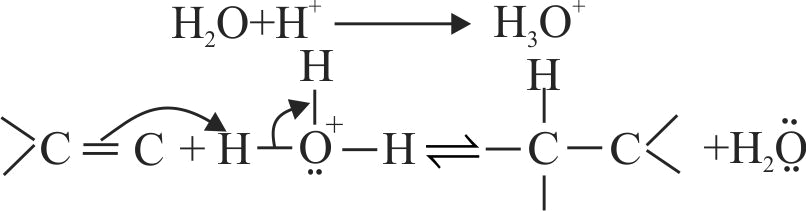
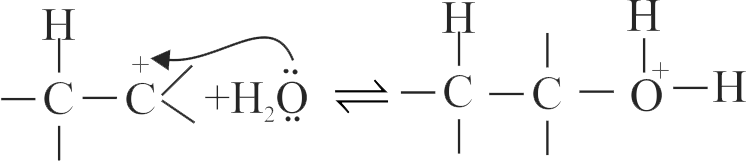
The acid catalysed hydration of alkene involves the following three steps.
I. Nucleophilic attack of water on carbocation.
II. Protonation of alkene to form carbocation by the electrophilic attack of \(\mathrm{H}_{3} \mathrm{O}^{+}\).
III. Deprotonation to form an alcohol.
Identify the sequence for the mechanism of reation in the acid catalysed hydration of alkenes.
322896
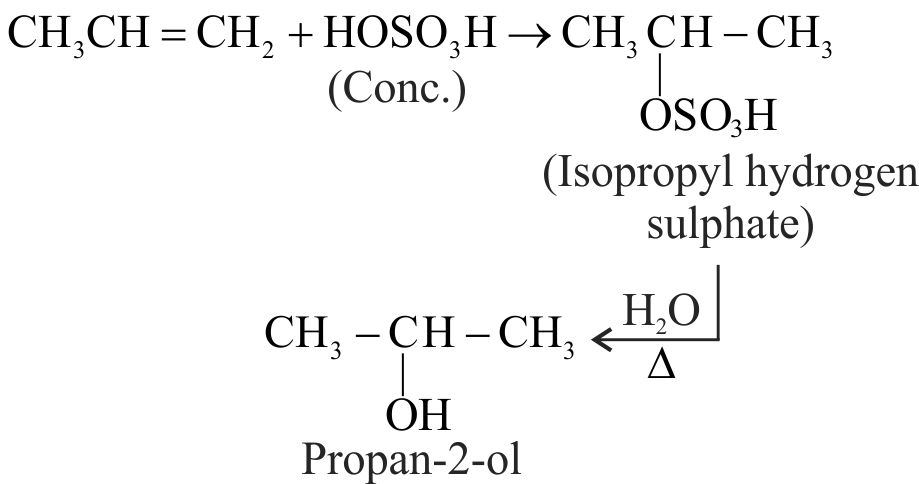
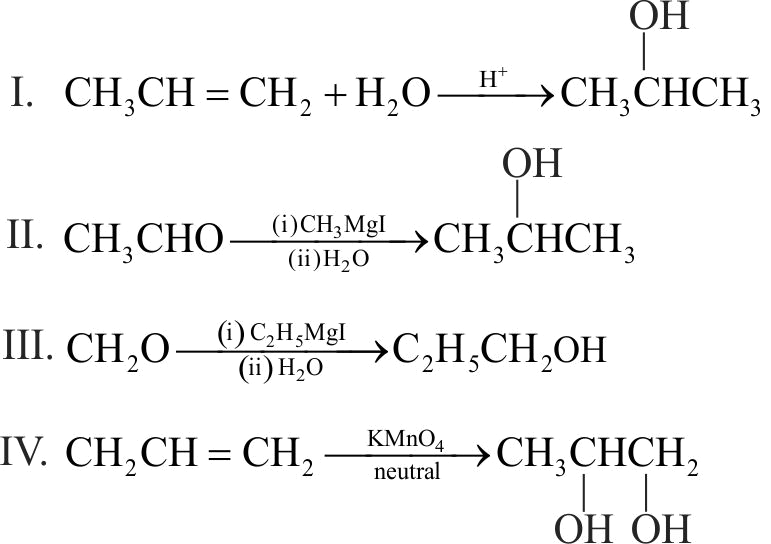
Which one/ones of the following reactions will yield 2-propanol?
I. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}\)
II. \({\text{C}}{{\text{H}}_{\text{3}}}{\text{ - CHO}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) C}}{{\text{H}}_{\text{3}}}{\text{MgI}}}}\)
III.\({\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{MgI}}}}\)
IV. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}\xrightarrow{{{\text{ Neutral KMn}}{{\text{O}}_{\text{4}}}}}\)
322894
The acid catalysed hydration of alkene involves the following three steps.
I. Nucleophilic attack of water on carbocation.
II. Protonation of alkene to form carbocation by the electrophilic attack of \(\mathrm{H}_{3} \mathrm{O}^{+}\).
III. Deprotonation to form an alcohol.
Identify the sequence for the mechanism of reation in the acid catalysed hydration of alkenes.
322896
Which one/ones of the following reactions will yield 2-propanol?
I. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}\)
II. \({\text{C}}{{\text{H}}_{\text{3}}}{\text{ - CHO}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) C}}{{\text{H}}_{\text{3}}}{\text{MgI}}}}\)
III.\({\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{MgI}}}}\)
IV. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}\xrightarrow{{{\text{ Neutral KMn}}{{\text{O}}_{\text{4}}}}}\)
322894
The acid catalysed hydration of alkene involves the following three steps.
I. Nucleophilic attack of water on carbocation.
II. Protonation of alkene to form carbocation by the electrophilic attack of \(\mathrm{H}_{3} \mathrm{O}^{+}\).
III. Deprotonation to form an alcohol.
Identify the sequence for the mechanism of reation in the acid catalysed hydration of alkenes.
322896
Which one/ones of the following reactions will yield 2-propanol?
I. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}\)
II. \({\text{C}}{{\text{H}}_{\text{3}}}{\text{ - CHO}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) C}}{{\text{H}}_{\text{3}}}{\text{MgI}}}}\)
III.\({\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{MgI}}}}\)
IV. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}\xrightarrow{{{\text{ Neutral KMn}}{{\text{O}}_{\text{4}}}}}\)
322894
The acid catalysed hydration of alkene involves the following three steps.
I. Nucleophilic attack of water on carbocation.
II. Protonation of alkene to form carbocation by the electrophilic attack of \(\mathrm{H}_{3} \mathrm{O}^{+}\).
III. Deprotonation to form an alcohol.
Identify the sequence for the mechanism of reation in the acid catalysed hydration of alkenes.
322896
Which one/ones of the following reactions will yield 2-propanol?
I. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}\)
II. \({\text{C}}{{\text{H}}_{\text{3}}}{\text{ - CHO}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) C}}{{\text{H}}_{\text{3}}}{\text{MgI}}}}\)
III.\({\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{MgI}}}}\)
IV. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}\xrightarrow{{{\text{ Neutral KMn}}{{\text{O}}_{\text{4}}}}}\)
322894
The acid catalysed hydration of alkene involves the following three steps.
I. Nucleophilic attack of water on carbocation.
II. Protonation of alkene to form carbocation by the electrophilic attack of \(\mathrm{H}_{3} \mathrm{O}^{+}\).
III. Deprotonation to form an alcohol.
Identify the sequence for the mechanism of reation in the acid catalysed hydration of alkenes.
322896
Which one/ones of the following reactions will yield 2-propanol?
I. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}{\text{ + }}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow{{{{\text{H}}^{\text{ + }}}}}\)
II. \({\text{C}}{{\text{H}}_{\text{3}}}{\text{ - CHO}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) C}}{{\text{H}}_{\text{3}}}{\text{MgI}}}}\)
III.\({\text{C}}{{\text{H}}_{\text{2}}}{\text{O}}\xrightarrow[{{\text{ (ii) }}{{\text{H}}_{\text{2}}}{\text{O}}}]{{{\text{ (i) }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}{\text{MgI}}}}\)
IV. \({\text{C}}{{\text{H}}_{\text{2}}}{\text{ = CH - C}}{{\text{H}}_{\text{3}}}\xrightarrow{{{\text{ Neutral KMn}}{{\text{O}}_{\text{4}}}}}\)